There’s no such thing as a vaccine generic in the way we think of generic pills. You can’t just copy a vaccine like you copy a cholesterol drug. Vaccines aren’t chemicals you can measure with a lab test. They’re living, complex biological products - made from viruses, bacteria, or mRNA instructions that teach your body to fight disease. That’s why you can’t slap a new label on a vaccine and call it generic. Every single batch has to be built from scratch, under tight control, with specialized tools and materials you can’t just order off Amazon.
Think about it this way: if a generic pill is like copying a printed page, a vaccine is like rebuilding a working airplane from scratch, using parts only three companies in the world make. That’s why, even though India produces 60% of the world’s vaccines by volume, the same companies that made the original vaccines - Pfizer, Moderna, AstraZeneca - still control the prices and the supply. And when a crisis hits, like the COVID-19 pandemic, the system breaks down fast.
Why vaccines can’t be copied like pills
Generic drugs work because they’re simple molecules. The FDA can check if a new version matches the original by testing its chemical structure, how fast it dissolves, and how it behaves in the body. That’s the ANDA process - Abbreviated New Drug Application. It cuts years off approval time.
Vaccines? No such shortcut. You can’t test bioequivalence because there’s no single molecule to measure. A vaccine is a whole system: live cells grown in bioreactors, purified with precision, mixed with stabilizers, packaged in vials that must stay cold from factory to arm. One mistake in temperature, one impurity in a lipid nanoparticle, and the whole batch fails. That’s why every vaccine needs a full Biological License Application - the same process the original maker used. No shortcuts. No savings. No fast track.
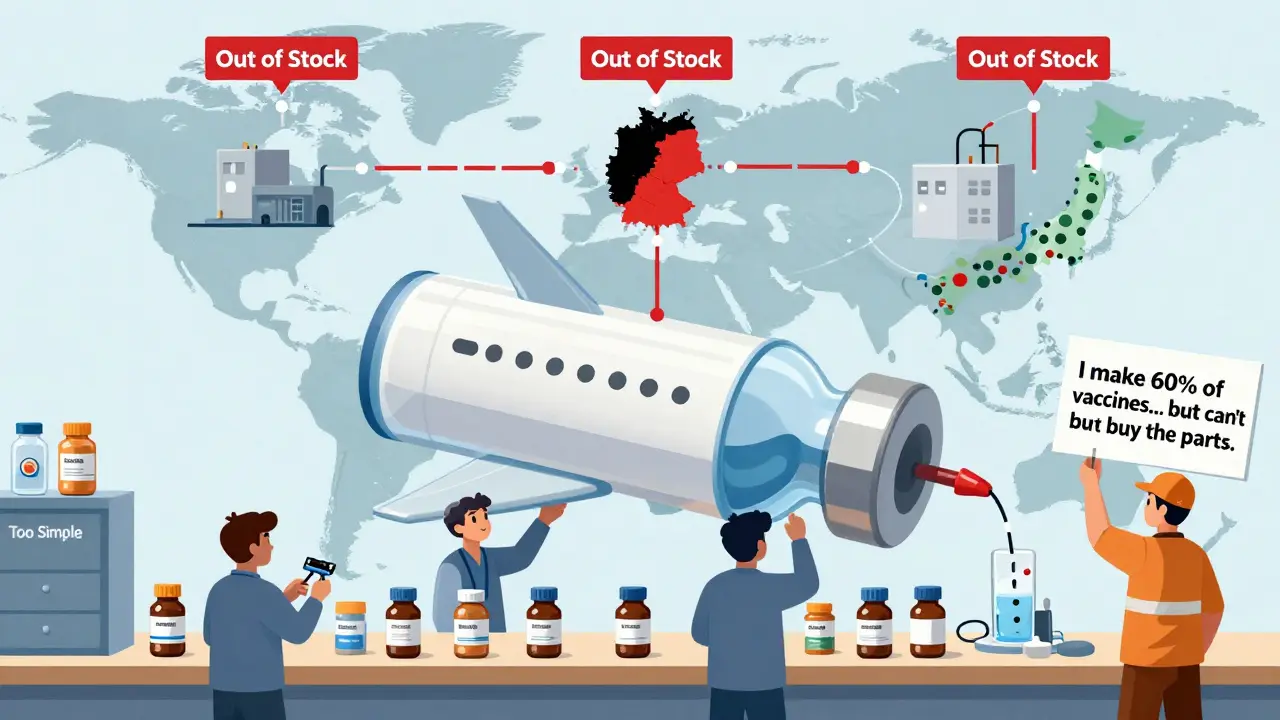
This isn’t theory. It’s fact. The WHO’s mRNA vaccine hub in South Africa, launched in 2021 with help from BioNTech, took 18 months just to start making doses. Not because the scientists didn’t know how - but because they couldn’t get the right tubing, filters, and lipid nanoparticles. Only five global suppliers make these materials. One factory in China shut down for a week in 2021, and vaccine production across Africa stalled.
The manufacturing gap: who builds, who gets
India makes more vaccines than any other country. The Serum Institute in Pune churns out 1.5 billion doses a year. It’s the backbone of global immunization - supplying 90% of the world’s measles vaccine, 70% of WHO’s DPT, and half of all COVID-19 doses given in low-income countries. Yet, it’s still a supplier, not a decision-maker.
When India’s second wave hit in April 2021, it stopped exporting vaccines. Not because it didn’t want to - but because hospitals in Delhi were running out. The world lost an estimated 50% of its vaccine supply overnight. That’s the reality of a system built on export dependency. India makes the doses, but doesn’t control the pricing, the patents, or the supply chains.
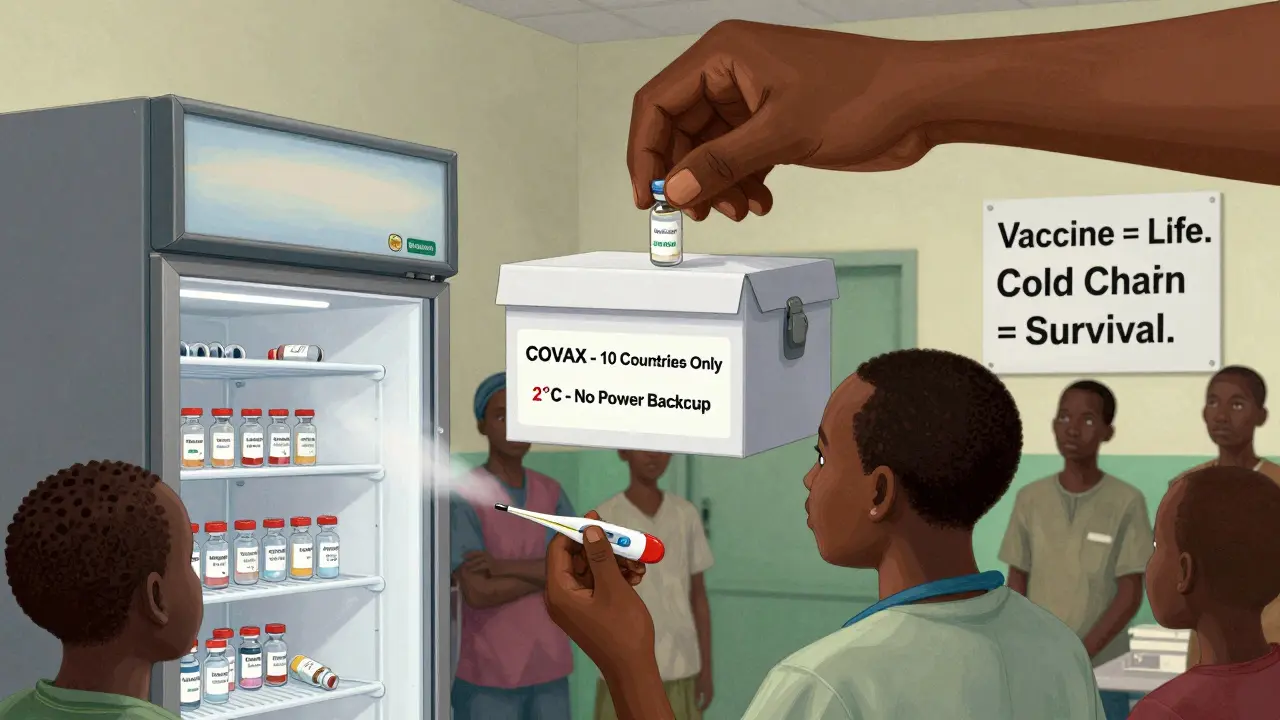
Meanwhile, high-income countries bought up 86% of the first COVID-19 doses. Just 16% of the world’s population, but 86% of the shots. In Africa, 23 countries had vaccinated less than 2% of their people by April 2021. Médecins Sans Frontières reported that 83% of the 1.1 million doses delivered to Africa through COVAX went to just 10 countries. The rest? Rotting in fridges with no cold chain to deliver them.
It’s not a shortage of will. It’s a shortage of infrastructure. In the Democratic Republic of Congo, health workers received doses expiring in two weeks. No refrigerated trucks. No trained staff. No backup power. A vaccine is useless if it can’t reach a child’s arm.
The cost of building capacity
Setting up a vaccine factory isn’t like opening a pharmacy. It takes 5 to 7 years and $200 million to $500 million. That’s not a startup cost - that’s a national investment. The African Union estimates it will take $4 billion and 10 years to get Africa making 60% of its own vaccines. Right now, Africa imports 99% of them.
Even India, with its 500 API manufacturers, imports 70% of its vaccine raw materials from China. That’s a vulnerability. When China restricted exports during the pandemic, Indian production dropped. The U.S. FDA warned in 2025 that overreliance on foreign manufacturing puts patient access at risk. Only 9% of API makers are in the U.S. - 44% are in India, 22% in China. But who controls the levers? The big five: GSK, Merck, Sanofi, Pfizer, and Johnson & Johnson. They made 70% of the $38 billion global vaccine market in 2020.
And pricing? It’s not competitive. In generic drugs, once five companies make the same pill, prices drop 80-90%. For vaccines? Not even close. Gavi, the Vaccine Alliance, says the pneumococcal vaccine still cost over $10 per dose for poor countries, even after promises of discounts. The Serum Institute made the AstraZeneca shot for $3-4 a dose - but that’s still barely above cost. The margins are razor-thin because the overhead is astronomical.

Technology transfer doesn’t work without materials
The WHO set up a technology transfer hub in South Africa to help low-income countries build their own mRNA vaccine lines. It’s a noble idea. But here’s the catch: you can’t transfer technology if you can’t get the materials. The lipid nanoparticles? The bioreactor membranes? The sterile filters? They’re made by a handful of firms in the U.S., Germany, and Japan. Export controls, supply shortages, and high prices block access.
Even if a country trains engineers, builds a clean room, and hires scientists, they’re still at the mercy of global suppliers. The hub in South Africa started producing in September 2023 - but only at 100 million doses a year. That’s less than 1% of global need. Meanwhile, the U.S. FDA launched a pilot in 2025 to speed up approval for generics made domestically - because they realized foreign inspections are slower, more expensive, and riskier.
The future: who holds the keys?
Dr. Soumya Swaminathan, former WHO Chief Scientist, pointed to three models for change: the Serum Institute of India, the small innovators like BioNTech and Moderna, and the University of Oxford. Each shows a different path - scale, speed, or open science. But none solve the core problem: who owns the materials, the machines, and the market?
Right now, the system is built on charity, not equity. Rich countries fund COVAX. Poor countries wait. India exports. Africa imports. The U.S. and Europe hoard. And the factories? They’re in the same hands they’ve always been.
There’s no magic fix. But there are clear steps: fund local manufacturing, lift export restrictions on raw materials, create global stockpiles of critical components, and force patent holders to license technology transparently. Until then, the idea of vaccine generics will remain a myth - not because the science is too hard, but because the system is too broken.
Why can’t we just make generic vaccines like we do with pills?
Vaccines are biological products, not chemical compounds. Unlike generic pills, which can be proven equivalent through simple lab tests, vaccines require full-scale manufacturing from scratch - including cell cultures, purification, and ultra-precise formulation. There’s no way to prove "bioequivalence" like with pills, so regulators require a full new license application for every vaccine, even if it’s nearly identical to the original.
Does India really make most of the world’s vaccines?
Yes. India produces about 60% of the world’s vaccines by volume, including 90% of the WHO’s measles vaccine and 70% of its DPT and BCG vaccines. The Serum Institute alone makes 1.5 billion doses annually. But despite this, India imports 70% of its vaccine raw materials from China, and its exports are often halted during domestic crises - like in 2021 during its COVID-19 surge.
Why do low-income countries still struggle to get vaccines?
It’s not just about production - it’s about access. High-income countries bought up most of the early COVID-19 doses. Many low-income nations lack cold chain infrastructure, trained staff, and stable supply chains. Even when doses arrive, they expire before use. In 2021, 23 African countries had vaccinated less than 2% of their populations, while 83% of Africa’s COVAX doses went to just 10 countries.
Can technology transfer solve vaccine inequality?
Technology transfer alone isn’t enough. The WHO’s mRNA hub in South Africa received training and blueprints from BioNTech, but still took 18 months to start production - mainly because it couldn’t source key materials like lipid nanoparticles. Without access to the raw components, even perfect blueprints won’t work. Manufacturing requires global supply chains, not just knowledge.
Why don’t more countries build their own vaccine factories?
It costs $200 million to $500 million and takes 5-7 years to build a single vaccine plant. Most countries can’t afford it. Even if they could, they’d still rely on foreign suppliers for critical materials. The African Union estimates it will take $4 billion and 10 years to reach 60% self-sufficiency - and that’s only if funding stays steady. Political will is often there, but capital and materials are not.








11 Comments
This is so important. We keep talking about vaccines like they're just pills with needles, but nope. It's a whole ecosystem. From the lipid nanoparticles to the cold chain to the factory floors. And yeah, India makes most of the doses but doesn't own the supply chain. That's not production parity. That's dependency. And it's unsustainable.
Let's stop pretending this is a science problem. It's a power problem.
As someone who's seen Serum Institute's factory up close I can tell you the truth is even more complicated. We don't just import raw materials from China we import the entire quality control protocol. The machines are American. The training is European. The standards are WHO but the enforcement? That's all us. We're the ones holding the line while the world watches. And when the pandemic hit and exports stopped? We didn't choose to hoard. We chose to save our own.
Why are we even having this conversation? The answer is simple. America built the mRNA tech. Europe funded the trials. China supplied the parts. India made the vials. Africa got the leftovers. That's not a system failure. That's capitalism. Stop romanticizing global equity. The world doesn't owe you a vaccine. You want one? Pay for it. Build it yourself. Or wait your turn.
Oh honey. You think this is about vaccines? Nah. This is about who gets to be the gatekeeper of life itself. The real story? Pfizer didn't invent mRNA. A German lab did. Moderna didn't patent it first. A Canadian uni did. But who got the billion-dollar contracts? The ones with the lobbyists. The ones with the FDA backchannels. The ones with the private jets. This isn't science. It's a monopoly dressed up as innovation.
The WHO hub in South Africa took 18 months because they were trying to build a nuclear reactor with Legos. You can't just hand someone a blueprint and expect them to make a vaccine. It's like giving a stranger a Tesla manual and asking them to build a battery. The materials are the bottleneck. Not the knowledge. And until we treat the supply chain like a strategic asset-not a commodity-we'll keep having the same crisis every time a factory in China shuts down for a week.
Did you know the lipid nanoparticles are made from a chemical that was originally developed for military nerve agents? And that the same company that makes them also supplies the U.S. military? Coincidence? Or is this all part of a plan to control population immunity through supply chain choke points? I've read the patents. The numbers don't add up.
Let me be perfectly clear: the notion that vaccines can be "generic" is not merely misguided-it is dangerously naive. Vaccines are not chemical entities; they are living, breathing, temperature-sensitive biological orchestrations. Each batch is a symphony of cells, lipids, and precision engineering. To equate them with ibuprofen is akin to comparing a Stradivarius to a ukulele. The regulatory frameworks, the manufacturing infrastructure, the supply-chain dependencies-all are fundamentally incompatible with the concept of "genericization." This is not a flaw in policy. It is a law of nature.
In India we say: "Jab tak khet nahi bana, tab tak khaana nahi milta." As long as the field isn't plowed, you won't get food. We've been the field for decades. But no one gave us the plow. We made 1.5 billion doses. But who decided where they went? Who decided the price? Not us. We're not the ones holding the keys. We're the ones holding the shovel.
Stop pretending this is a moral issue. It's an economic one. No one will build a $500M factory for $3/dose profit margins. Period. The market doesn't reward altruism. It rewards control. And right now, control sits with five companies. Fix the profit model or stop crying about equity.
I appreciate the depth of this analysis. What strikes me most is the quiet dignity of the workers in Pune, Johannesburg, and Dhaka who show up every day to do the work that the world depends on-but never sees. The system is broken, yes. But the people? They're not broken. They're the ones holding it together. Maybe the solution isn't just about patents or pipelines. Maybe it's about dignity. Recognition. Fairness. Not as charity. As right.
The cold chain isn't just about refrigeration-it's about systemic resilience. We've seen it in Johannesburg: doses expiring because the last-mile transport failed. No backup generator. No trained nurses. No logistics support. Tech transfer? Great. But without infrastructure, it's just data on a USB. We need sovereign capacity-not just blueprints. And we need it yesterday. 🚨💉