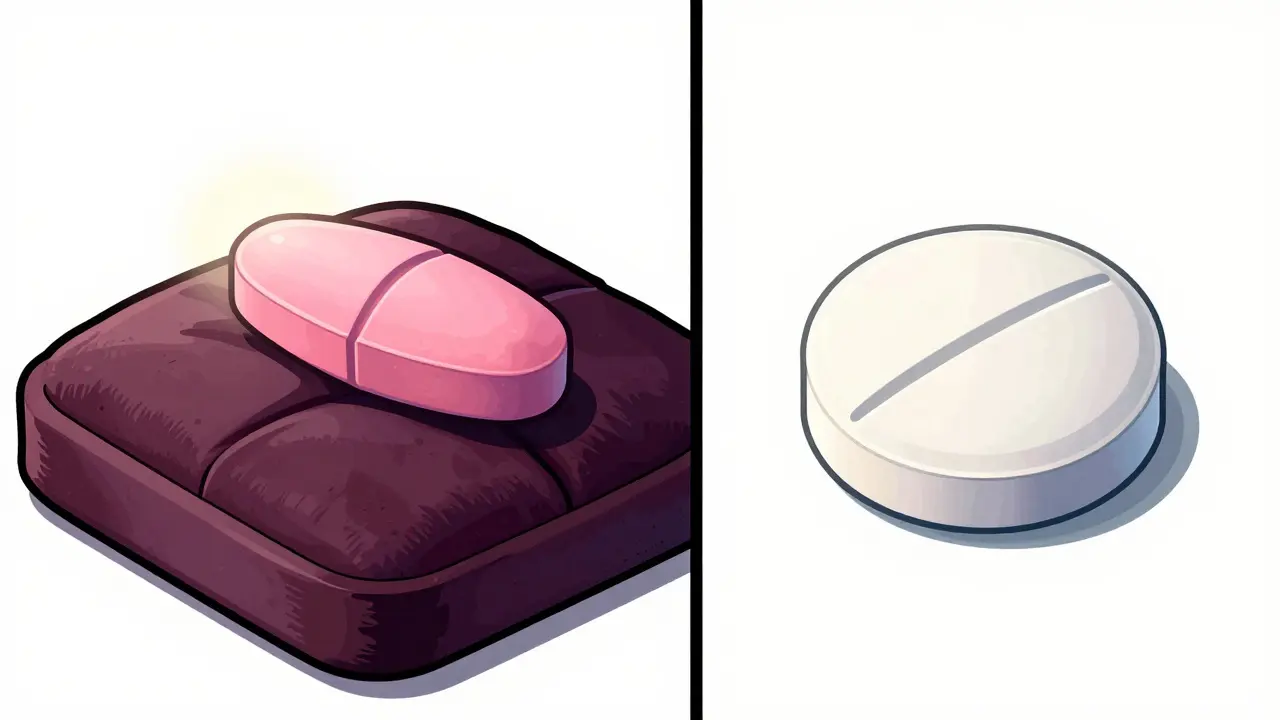
The short answer is yes, the active part is the same. However, the inactive ingredients-the stuff that makes up the rest of the pill-can vary wildly between brands. While these components don't treat your illness, they change how your body interacts with the pill. For 98% of people, this is a non-issue. But for a small group, these "invisible" ingredients can make a world of difference in how a medication feels.
What are inactive ingredients and what do they actually do?
In the pharmaceutical world, inactive ingredients are called excipients. Excipients are non-pharmaceutical components that don't have a therapeutic effect but are essential for the drug's delivery and stability. Think of them like the flour and eggs in a cake; the sugar is the part you're after, but without the rest, you just have a pile of crystals.
These ingredients serve several technical roles. Some act as fillers to make a pill large enough to actually hold in your hand, while others are binders that stop the tablet from crumbling into powder the moment you touch it. Some are disintegrants, which are designed to help the pill dissolve at exactly the right speed in your stomach. There are also coatings that make a bitter pill easier to swallow or preservatives like sodium benzoate that keep the medication from spoiling on the shelf.
The FDA's rules on generic vs. brand-name drugs
When a company wants to release a generic version of a drug, the U.S. Food and Drug Administration (FDA) requires it to be bioequivalent. This means the generic must deliver the same amount of active ingredient into your bloodstream at about the same rate as the brand-name version.
The FDA allows a small amount of wiggle room. While the gold standard is a 1:1 match, the accepted range for variation is generally between 80% and 125%. In reality, most generics are much tighter than that, with an average difference of only about 4%. Because of this, the therapeutic effect remains the same for the vast majority of patients.
However, the FDA does not require the inactive ingredients to be the same. This is why your brand-name pill might be a pink oval, but your generic is a white circle. Trademark laws actually force them to look different, and different manufacturers use different "recipes" for their fillers and binders.
| Feature | Brand-Name Drug | Generic Drug |
|---|---|---|
| Active Ingredient | Same | Same |
| Inactive Ingredients | Specific Proprietary Blend | Varies by Manufacturer |
| FDA Bioequivalence | The Benchmark | Must match benchmark (approx. 4% variance) |
| Appearance | Unique/Trademarked | Must be different from brand |
| Average Cost | Higher (Often 80-85% more) | Significantly Lower |

Why some people react differently to generics
If you're among the 1-2% of people who feel a difference when switching to a generic, it's usually not the drug itself causing the issue-it's a sensitivity to an excipient. For example, lactose is a common filler used in about 40% of oral medications. If you have severe lactose intolerance, a generic that uses lactose as a filler might cause gastrointestinal distress, even if the brand-name version didn't.
Other common culprits include dyes, sulfites (often found in injectables), or soya lecithin. You might experience mild nausea, a skin rash, or a change in how quickly the drug "kicks in" if the disintegrant in the generic dissolves slower than the one in the brand-name version.
There is also a specific category of drugs called Narrow Therapeutic Index (NTI) drugs. These are medications where a tiny change in the dose or absorption can lead to a big change in how the drug works. Examples include levothyroxine (for thyroid) or warfarin (a blood thinner). For these specific meds, even a 4% difference in absorption can occasionally push a patient's levels out of the safe zone, requiring a switch back to the brand name or a different specific generic manufacturer.
How to manage your switch to a generic
Switching to a generic is usually a financial win-saving hundreds of dollars a month on drugs like atorvastatin is a huge relief for most. But if you have a history of allergies or are taking an NTI drug, you can take a few proactive steps to ensure the transition is smooth.
- Request the ingredient list: Ask your pharmacist for the "package insert" or the list of inactive ingredients for both the brand and the generic.
- Keep a medication diary: For the first 2-4 weeks after switching, jot down any new symptoms. Note the time of day and any GI issues. This helps your doctor determine if a side effect is a real reaction or just a coincidence.
- Consistency is key: If you find a generic that works well, try to stick with that specific manufacturer. Because different generics can have different inactive ingredients, switching from "Generic A" to "Generic B" can sometimes feel like a whole new medication switch.
- Ask about Authorized Generics: Some companies offer Authorized Generics. These are generic versions made by the original brand manufacturer, meaning they use the exact same inactive ingredients as the brand name but at a lower price.
Is it possible for a generic to be less effective?
In terms of the active drug, no. The FDA requires generics to be bioequivalent, meaning they provide the same therapeutic effect. However, if you are sensitive to a specific filler or if the drug has a narrow therapeutic index, you might perceive a difference in efficacy due to how your body absorbs that specific formulation.
Why does my generic pill look different from my brand-name pill?
Trademark laws prevent generic manufacturers from copying the exact look, shape, and color of a brand-name drug. This is why you'll see different colors or shapes, even though the medicine inside is the same.
What should I do if I think my generic is causing side effects?
First, don't stop taking your medication abruptly. Contact your pharmacist or doctor and tell them you suspect a reaction to the inactive ingredients. They can help you identify the specific excipient causing the problem and potentially switch you to a different generic manufacturer that doesn't use that ingredient.
Are there any generics that are "cleaner" than others?
There is a growing trend toward "clean label" pharmaceuticals that use fewer dyes and fillers. While not yet standard for all prescriptions, more manufacturers are moving toward allergen-free formulations to accommodate patients with sensitivities.
Does a lower price mean lower quality?
No. The price is lower because generic companies don't have to spend billions on the original research, clinical trials, and marketing that the brand-name company did. They simply have to prove to the FDA that their version is bioequivalent.
Next steps for a safe transition
If you're currently feeling a difference after a switch, your first move should be to check the inactive list for common allergens like lactose or gluten. If you're on a high-stakes medication for your heart or thyroid, ask your doctor to monitor your blood levels more frequently during the first month of the switch. Most people will find that generics work perfectly, but being aware of the "hidden" ingredients helps you take control of your own health outcomes.